π-interactions
π-interactions are types of non-covalent interactions that involve the attractive forces between ions or molecules and the electron-rich π-systems of aromatic rings.
TODO:
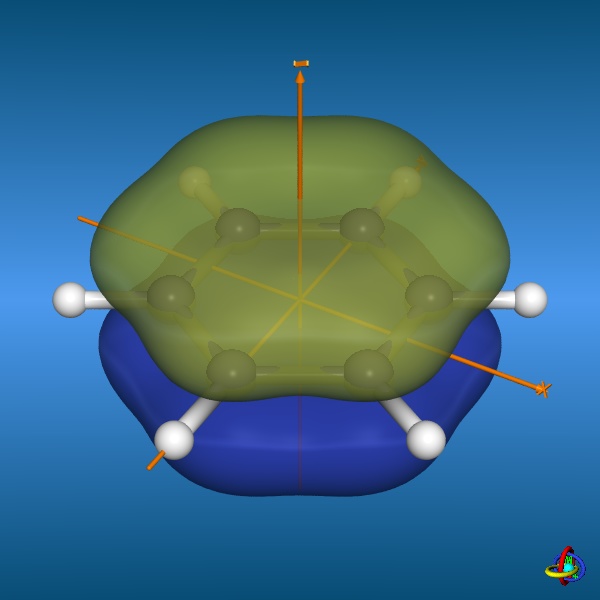
=== “Benzene lowest-occupied molecular orbital”
<figure markdown>
{ alight=left width=300 }
</figure>
**Credit:** [Dr. S. Immel](http://csi.chemie.tu-darmstadt.de/ak/immel/tutorials/orbitals/molecular/benzene.html#orbitals)
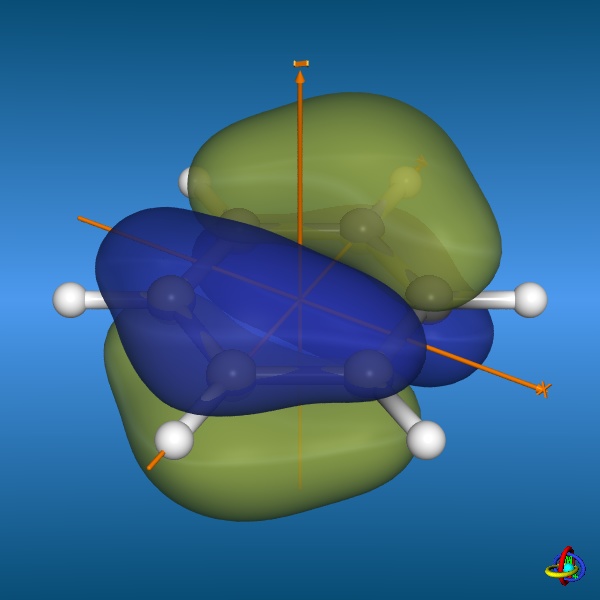
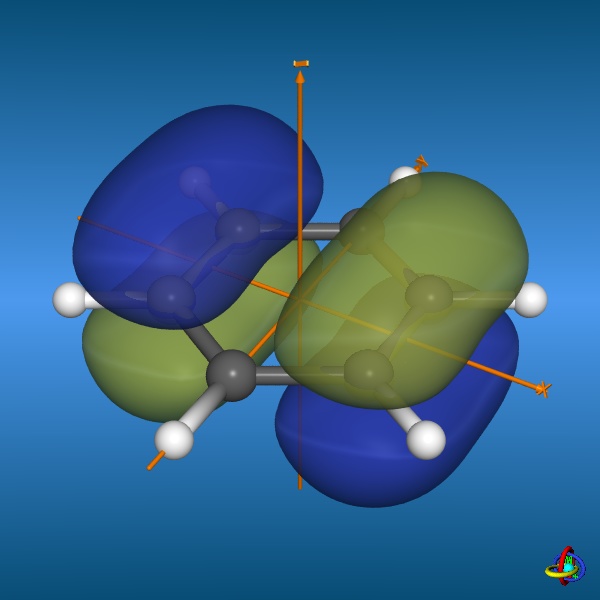
=== “Benzene highest-occupied molecular orbitals”
<figure markdown>
{ alight=left width=300 }
</figure>
<figure markdown>
{ alight=left width=300 }
</figure>
**Credit:** [Dr. S. Immel](http://csi.chemie.tu-darmstadt.de/ak/immel/tutorials/orbitals/molecular/benzene.html#orbitals)
π-π
=== “Edge-to-face”
<figure markdown>
{ alight=left width=200 }
</figure>
=== “Displaced”
<figure markdown>
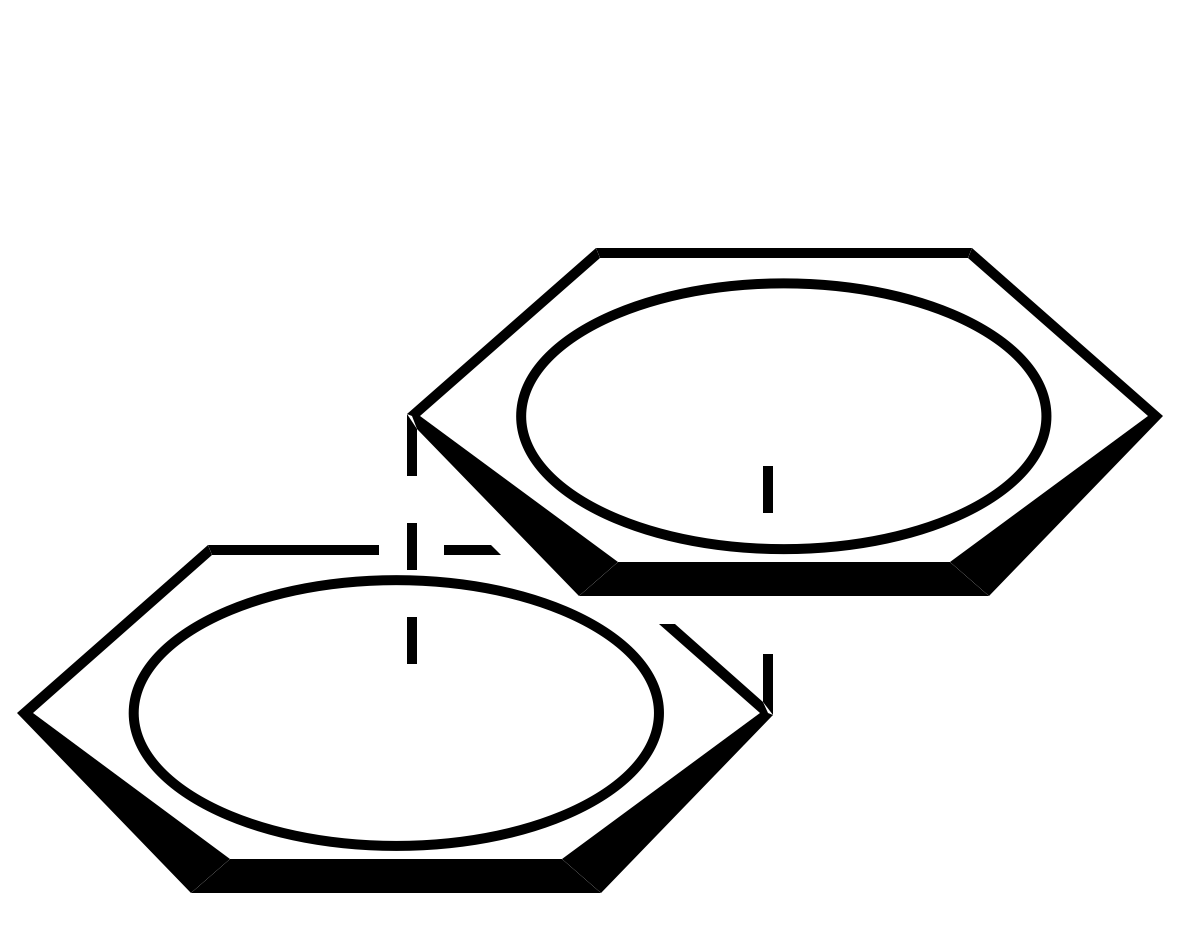{ alight=left width=200 }
</figure>
=== “Face-to-face”
<figure markdown>
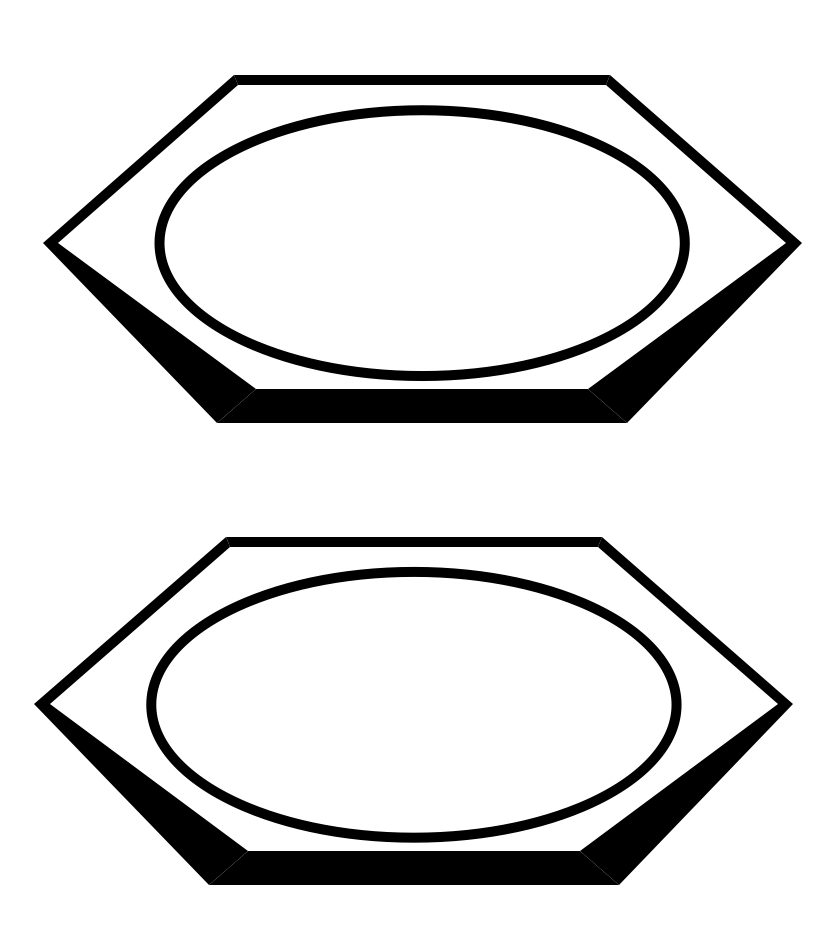{ alight=left width=200 }
</figure>
Cation-π
Last updated on